Updated 20 Feb 2020
The latest infectious disease sweeping the globe, the novel coronavirus, has now infected tens of thousands of people. While most of the affected people are in China, the virus has been popping up in cities around the world.
While there have been other types of coronavirus before – think SARS that broke out in 2002-3, or MERS in 2012 – this particular type is new. First spotted in Wuhan, China, scientists think it originated in an animal and spread to humans, then from human to human.
With the rate of travel and global trade today, and the tendency of viruses to rapidly mutate, it can be hard to tell how much of an impact a specific virus might have on the world. Scientists are scrambling to understand this new virus and its behaviour at lightspeed so they can develop, test and produce a new vaccine to protect us.
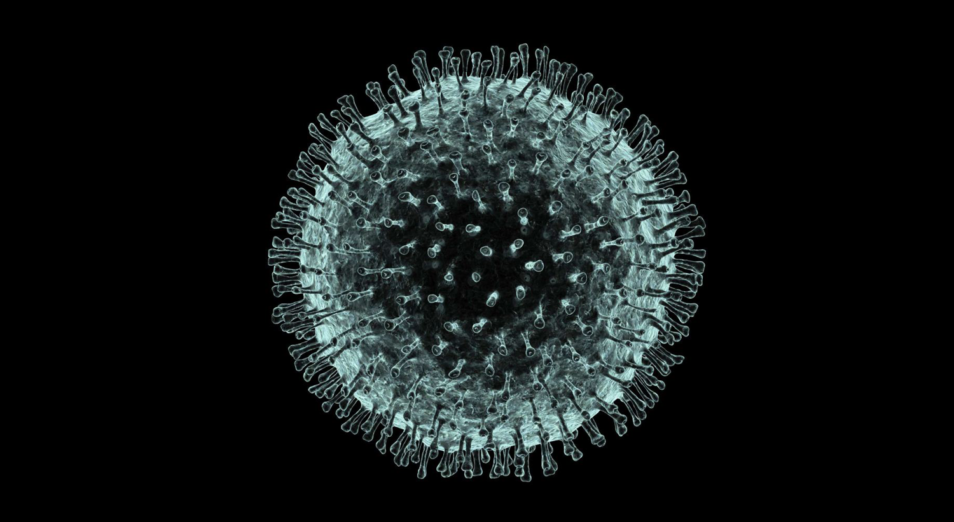
Computer artwork of the new coronavirus, named after its distinctive ‘crown’ of surface proteins. Science Photo Library: PASIEKA
Creating a coronavirus vaccine in months, rather than years
Vaccine development is complicated and lengthy. Traditionally it takes months to years to take a vaccine from development out into the world. It’s a long process of developing possible vaccines, pre-clinical testing, human trials, regulatory approval and large-scale development.
But this isn’t the first time the world has faced down an epidemic. Our own scientists have been heavily involved in infectious disease work. This includes understanding how SARS spreads, tracking the spread of avian flu, developing the Hendra vaccine, and much more. Research organisations have been down this path before, and learned that the best way to speed up the response is to be really, really prepared.
That’s why we’re part of a global alliance: the Coalition for Epidemic Preparedness Innovations (CEPI). It aims to derail epidemics by speeding up the development of vaccines. Within the coalition we’re involved in various parts of a rapid vaccine development ‘pipeline’. Each of our organisations form a part of the pipeline, ready to go when an epidemic hits. This way vaccines can be developed, tested and delivered much faster than ever before.
What we’re doing – a team effort
First up, we have sourced a sample of the novel coronavirus – now known as the virus responsible for COVID-19. This sample is from the Peter Doherty Institute for Infection and Immunity, who were the first organisation outside of China to re-create the virus in the lab. This sample has gone to our high-containment facility in Geelong, the Australian Centre for Disease Preparedness (ACDP).
Our highly trained scientists are working in this secure facility to better understand the virus. We’re exploring: how does it behave? How does it infect and spread? And what are its characteristics?

We are working on coronavirus in the secure area of our Australian Animal Health Laboratory in Geelong.
Once we’ve figured that out, our team at AAHL will start pre-clinical testing of new potential vaccines. This includes potential vaccines from the University of Queensland who are already hard at work.
If those tests show a vaccine is successful, other research organisations will be able to move on to full human trials of the vaccine.
We also have advanced manufacturing capabilities that could create the proteins other organisations need in developing new vaccine candidates.
It’s truly a global team effort in response to a global challenge, and the scientific community is moving at unprecedented speed. If all goes well, the world could be looking at a new vaccine in months rather than the usual years. Find out more about how long it will be before a vaccine against the coronavirus is publicly available.


14th August 2020 at 5:31 pm
Many countries are racing against the clock to find the cure to this new global pandemic, and what could be the fastest way but understanding this virus. Thanks for this great article.
1st April 2020 at 11:08 pm
GO HARD , ;LADIES AND GENTS , OF THE CSIRO / DOHERTY INSTIUTE , I HOPE THE PRIME MINISTERS AND GOVERNMENT ADVISERS , THAT STRIPPED BACK YOU FUNDING, ARE SQUIRMING IN EXTREME DISCOMFORT, TO SAY THE LEAST!