
Sustainable farming can reduce nitrous oxide emissions. eutrophication&hypoxia/Flickr
Nitrous oxide (N₂O) (more commonly known as laughing gas) is a powerful contributor to global warming. It is 265 times more effective at trapping heat in the atmosphere than carbon dioxide and depletes our ozone layer.
Human-driven N₂O emissions have been growing unabated for many decades, but we may have been seriously underestimating by just how much. In a paper published today in Nature Climate Change, we found global emissions are higher and growing faster than are being reported.
Although clearly bad news for the fight against climate change, some countries are showing progress towards reducing N₂O emissions, without sacrificing the incredible crop yields allowed by nitrogen fertilisers. Those countries offer insights for the rest of the world.
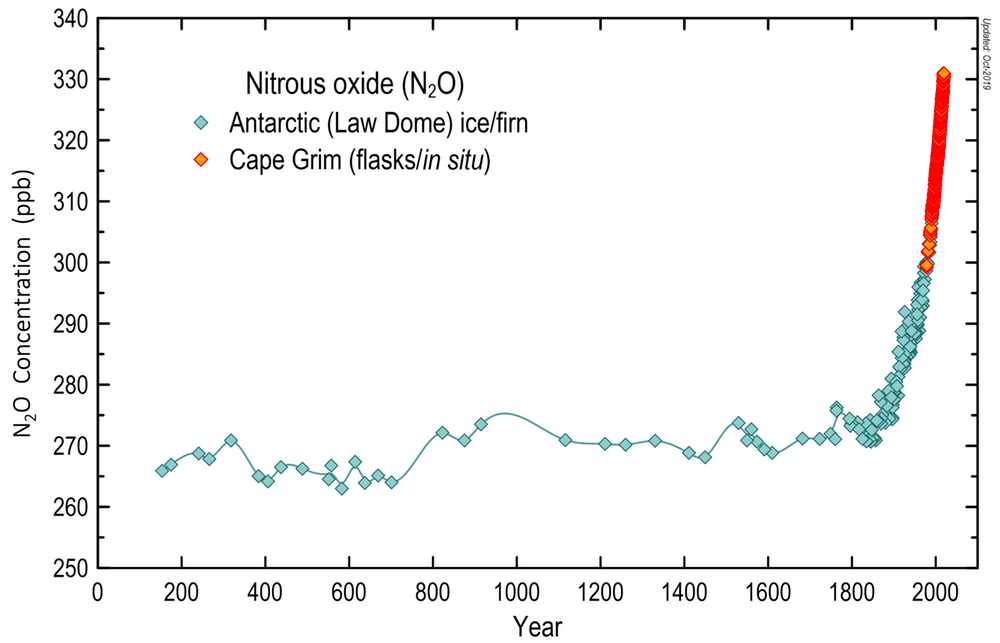
N₂O concentrations (parts per billion) in air from Cape Grim Baseline Air Pollution Station (Tasmania, Australia) and air contained in bubbles trapped in firn and ice from the Law Dome, Antarctica. N₂O concentrations from these two sites reflect global concentrations, not local conditions. Source: BoM/CSIRO/AAD.
The Green Revolution
There are a number of natural and human sources of N₂O emissions, which have remained relatively steady for millennia. However, in the early 20th century the Haber-Bosch process was developed, allowing industry to chemically synthesise molecular nitrogen from the atmosphere to create nitrogen fertiliser.
This advancement kick-started the Green Revolution, one of the greatest and fastest human revolutions of our time. Crop yields across the world have increased many times over due to the use of nitrogen fertilisers and other improved farming practices.
But when soil is exposed to abundant nitrogen in its active form (as in fertilizer), microbial reactions take place that release N₂O emissions. The unrestricted use in nitrogen fertilisers, therefore, created a huge uptick in emissions.
N₂O is the third-most-important greenhouse gas after carbon dioxide and methane. As well as trapping heat, it depletes ozone in the stratosphere, contributing to the ozone hole. Once released into the atmosphere, N₂O remains active for more than 100 years.
Tracking emissions from above
Conventional analysis of N₂O emissions from human activities are estimated from various indirect sources. This include country-by-country reporting, global nitrogen fertiliser production, the areal extent of nitrogen-fixing crops and the use of manure fertilisers.
Our study instead used actual atmospheric concentrations of N₂O from dozens of monitoring stations all over the world. We then used atmospheric modelling that explains how air masses move across and between continents to infer the expected emissions of specific regions.
We found global N₂O emissions have increased over the past two decades and the fastest growth has been since 2009. China and Brazil are two countries that stand out. This is associated with a spectacular increase in the use of nitrogen fertilisers and the expansion of nitrogen-fixing crops such as soybean.
We also found the emissions reported for those two countries, based on a methodology developed by the Intergovernmental Panel on Climate Change, are significantly lower than those inferred from N₂O levels in the atmosphere over those regions.
This mismatch seems to arise from the fact that emissions in those regions are proportionally higher than the use of nitrogen fertilizers and manure. This is a departure from the linear relationship used to report emissions by most countries.
There appears to be a level of nitrogen past which plants can no longer effectively use it. Once that threshold is passed in croplands, N₂O emissions grow exponentially.
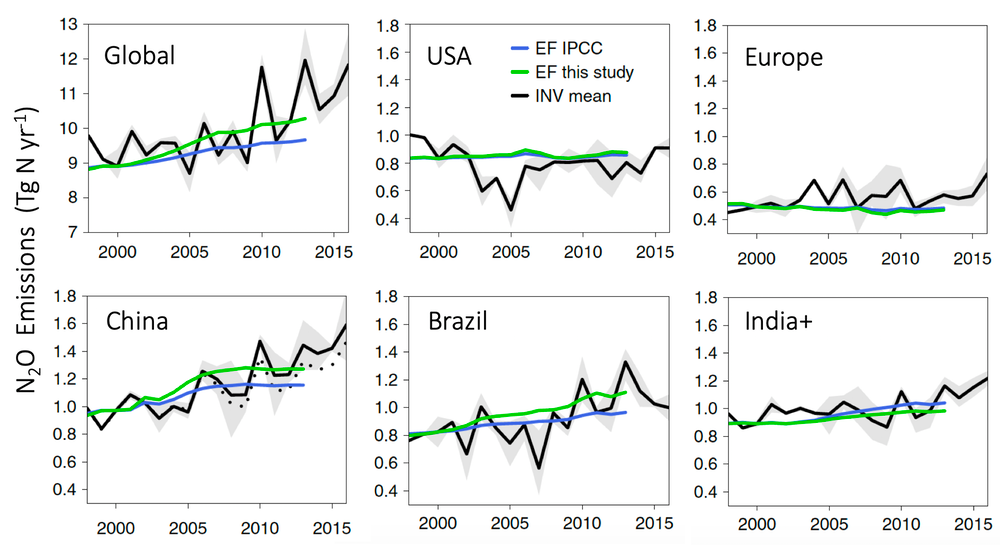
N₂O emissions from agriculture estimated by using the emissions factors approach of the IPCC (blue), the calculated emission factor in this study (green), and the average of the atmospheric inversions in this study (black). Source: Thompson et al. 2019 Nature Climate Change
Reversing the trends
Reducing N₂O emissions from agriculture will be very challenging, given the expected global growth in population, food demand and biomass-based products including energy.
However, all future emission scenarios consistent with the goals of the Paris Agreement require N₂O emissions to stop growing and, in most cases, to decline – between 10% and 30% by mid-century.
Interestingly, emissions from the USA and Europe have not grown for over two decades, yet crop yields across these regions increased or remained steady. Both regions have created strong regulations largely to prevent excess accumulation of nitrogen in soils and into waterways.
These areas and other studies have demonstrated the success of more sustainable farming in reducing emissions while increasing crop yields and farm-level economic gains.
A whole toolbox of options is available to increase nitrogen use efficiency and reduce N₂O emissions: precision applications of nitrogen in space and time, the use of N-fixing crops in rotations, reduced tillage or no-tillage, prevention of waterlogging, and the use of nitrification inhibitors.
Regulatory frameworks have shown win-win outcomes in a number of countries. With intelligent adaptions to different nations’ and regions’ needs, they can also work elsewhere.![]()
This article is republished from The Conversation under a Creative Commons license. Read the original article.


27th November 2019 at 12:50 am
the real problem with agricultural fertilizer is the runoff into the groundwater, streams rivers , esturaries where they favor abnormal growth of cyanobacteria , which not being part of the food chain, die, and decompose. This eats up all the oxygen in our productive rivers , lakes , estuaries oceans killing sea life…. everywhere…. Add this to the overfishing problem… and you have a real crisis that the article forgot to mention…. why is it that I don’t trust the government, nor the media.?
26th November 2019 at 6:58 am
More reasons to get biochar rolled out. Biochar can reduce nitrogen use.
20th November 2019 at 3:16 pm
I am being picky again, but the Haber-Bosch Process does not synthesise molecular nitrogen. It makes ammonia. Otherwise, a valuable article. Let’s hope farmers everywhere can have some means of measuring phosphate and nitrogen content of their farmland, plus knowledge that there is a point where more is not better.