Part of the Biosecurity Series
By John Lowenthal and Andrew Bean
Zoonoses are diseases that have the ability to spread from animals to people, and they include some very well known diseases such as tuberculosis, flu and rabies, as well as some less familiar newcomers such as the Nipah and Melaka viruses.
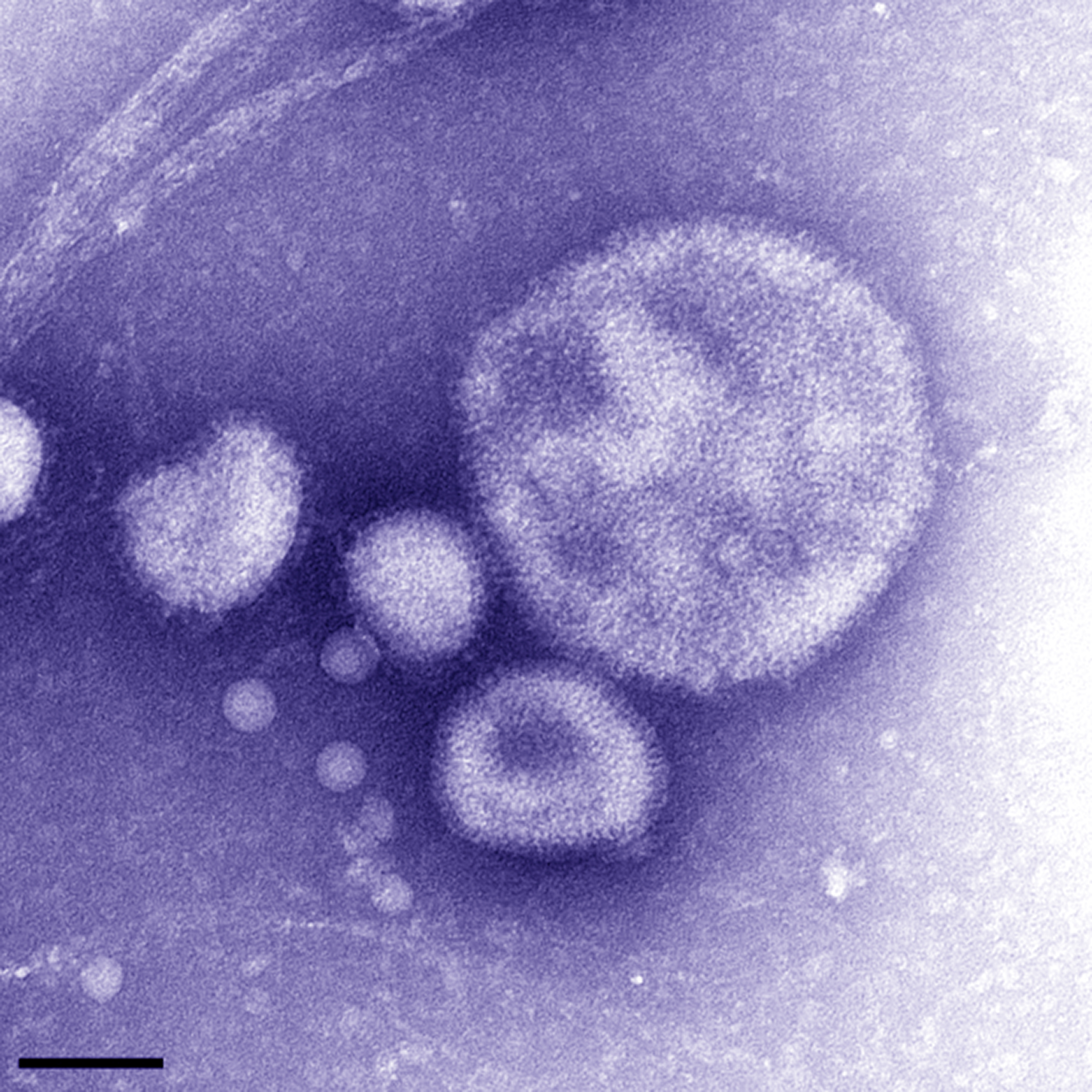
Mug shot of a one of the world’s new deadly viruses, A(H7N9).The four blobs in this electron micrograph are the virus.
In recent times zoonoses have accounted for more than 70 per cent of all emerging diseases, including H7N9 and H5N1 avian influenza, SARS, and MERS. What’s interesting is that a great deal of these zoonotic viruses that now pose a problem for humans appear to originate in either bats or poultry.
This highlights our need to understand not just what is happening in the human, but also what is happening in the animal. Wild animals such as bats and migratory water birds are the natural ‘reservoir’ hosts for many zoonotic infections and little is known about how they carry these viruses without showing signs of disease.
Other animals, including horses, pigs, chickens and even people are ‘spillover’ hosts, meaning they are highly susceptible to these viruses, and infection is usually deadly.
The recent growth and geographic expansion of human populations and the advance of agriculture into wildlife habitats has meant that now, more than ever, there is a greater risk of emerging infectious diseases being transmitted to people from wild and domesticated animals.
In addition, the impact of climate change has resulted in disturbances in eco-systems and a re-distribution of disease hosts and carriers. Increased global travel means a greater likelihood that new infectious agents will rapidly spread amongst the human population.
The World Health Organization has warned that the source of the next human pandemic is likely to be zoonotic, and that wildlife is a prime culprit. While the current list of known emerging infectious diseases is a major concern, it is the unknown virus lurking out there, with a potential for efficient human to human transmission that may pose the biggest pandemic threat.
A rapidly spreading lethal airborne zoonotic virus would, of course, be a major concern. You may remember the 2011 movie Contagion, which showed a fast-moving epidemic and the struggle to find a cure and control the panic. The ABC’s Catalyst story Virus Hunters also demonstrates the threat of quickly spreading diseases, and looks at the research our scientists do in the high containment facilities at the Australian Animal Health Laboratory.
If we want to fight these emerging threats and come out on top, we need to take a different approach to what we have done in the past and integrate medical, veterinary, ecological and environmental research.
This is what we refer to as the One Health approach – a combined approach to animal, human and environmental health, and the idea that we can all benefit from working together to value and solve the health problems of the world and reduce the risk of the next pandemic.
We believe it’s important to study and compare the disease in both the natural and spillover hosts. For example, understanding the differences between the immune systems of domesticated and wild animal hosts and comparing them to people is crucial for identifying the underlying disease mechanisms involved in zoonotic infections, and for developing new strategies for disrupting their transmission to humans.
This has important implications for predicting, preventing and controlling spillover events, and for the development of new therapeutics, vaccines and diagnostics.

The Australian Animal Health Laboratory in Geelong, Victoria
Improving knowledge, prevention and treatment of zoonoses is the focus of the One Health research that we’re undertaking with our national and international partners, and within our unique high containment facility at AAHL– the world’s most sophisticated high containment facility. Focusing our research efforts in this area will assist in facilitating the development and application of effective and sustainable community health strategies. There is a growing view that a One Health approach will be critically important for our preparedness for the next zoonotic pandemic.
Join the Conversation: #bflaunch
About the Authors
 John Lowenthal is Theme Leader for A One-Health approach to Emerging Infectious Diseases, CSIRO Biosecurity Flagship
John Lowenthal is Theme Leader for A One-Health approach to Emerging Infectious Diseases, CSIRO Biosecurity Flagship
John’s research is in the area of veterinary health and immunology, including studying the innate immune responses to viral diseases, assessing the ability of immune modulators such as cytokines to enhance resistance to disease and improve vaccine efficacy, using RNA interference to modulate disease-resistance, development of novel therapeutics for zoonotic viruses (H5N1 flu, Hendra virus) and the development of disease-resistant animals.

Andrew Bean is Stream Leader for Animal Biosecurity, CSIRO Biosecurity Flagship.
Andrew is an immunologist working to improve animal and human health with a ‘One Health’ approach. He joined CSIRO’s Australian Animal Health Laboratory in 1998 and the emphasis of his work is now on the innate immune response and the therapeutic and immuno-enhancing qualities of cytokines with the potential to improve health. His current research areas include avian influenza, Hendra virus, immune molecules and receptors, developing and assessing antiviral therapy, vaccines and adjuvants and therapeutics.