With the rise of electric cars and energy storage, graphite – a mineral made of carbon – is poised to be hot on the commodity market to meet demand for lithium-ion batteries.
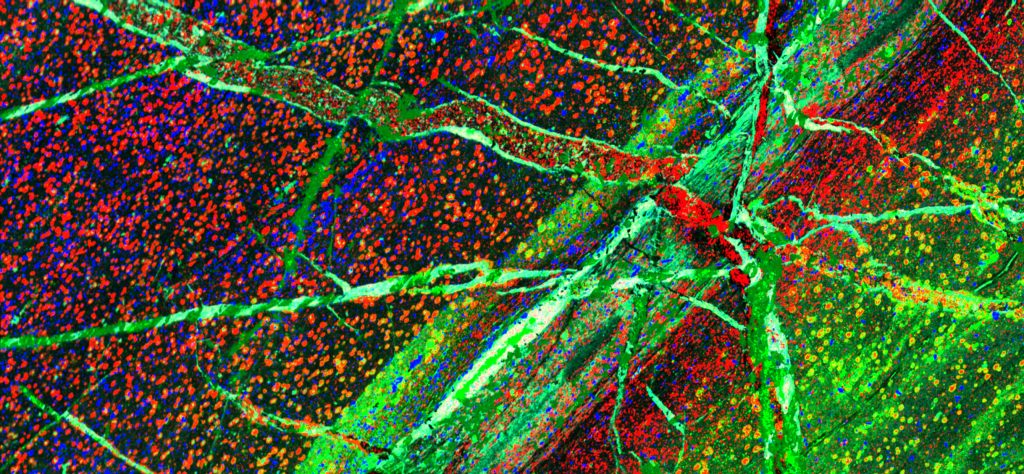
Graphite up close: A 3D graphite sample structure merging into a colourful 2D x-ray map highlighting the chemistry within a graphite deposit.
With the rise of electric cars and energy storage, graphite – a mineral made of carbon – is poised to be hot on the commodity market to meet demand for lithium-ion batteries.
A mineral of extremes, graphite is the strongest and stiffest naturally occurring material, while contrastingly soft and lightweight. It’s also heat resistant with a high melting point, similar to that of a diamond. These properties, coupled with high conductivity, make graphite critical for use in batteries.
Positively charged demand
Specifically, graphite is needed for battery anodes, the positively charged electrode through which the current flows. While graphite has been used as a key ingredient in anodes in all kinds of batteries for decades, lithium-ion batteries contain proportionately about double the amount of graphite.
To put this into perspective, there is estimated to be 54kg of graphite in the batteries used in each Tesla Model S (85 kWh).
But, not all graphite is equal.
Graphite comes naturally and abundantly in three different forms: ‘crystal’ flake, lump and amorphous. Only high purity (>99.9 per cent) graphite flake is capable of producing the level of electrical conductivity needed to be considered ‘battery-grade’.
About one million tonnes of the mineral is sold annually worldwide. Most of the world’s graphite supply comes from China and Brazil, so a lot of work is going into diversifying the sources of graphite to meet the increasing demand predicted for the future.
Companies seeking to get a competitive edge in this market, are finding ways to improve their production processes so that they can recover more graphite flake, at the highest level of purity possible.
Importance of understanding how graphite forms
Graphite is a metamorphic mineral that has undergone transformation deep underground as a result of heat, pressure and flowing fluid. Graphite flake crystals grow with increasing temperature – upwards of 750 degrees Celsius – as organic matter such as dead bacteria or plants in younger rocks is “cooked up” and converted to graphite.
These geological processes lead to mineral ‘rich zones’ and variability within a deposit.
That’s why understanding the geological history of a graphite deposit is crucial for determining the quality of the mineral contained and how a company can best mine and process it to maximise the value recovered.
Getting ahead in the graphite market
Our researchers are applying analysis techniques to characterise rock samples, helping to reveal the history, geology and variability of mineral deposits. Working with ASX-list Talga Resources, we revealed how the world’s highest grade and extremely conductive graphite deposit, Nunasvarra, was formed about 1.8 billion years ago.
Talga’s Nunasvarra is the largest graphite deposit in Europe and contains so much graphite that the rocks conduct enough electricity to complete a circuit with a battery and lightbulb.
The company aims to turn the graphite into graphene, which fetches much higher prices. To do this, they need to be able to exfoliate the graphite rock into individual one-atom-thick sheets of graphene. By characterising the geology of Nunasvarra, we could reveal what other minerals it contained that could impede or improve Talga’s production process method for graphite and graphene.
In another similar project, we worked with ASX-listed company, Kibaran Resources, to shed light on their graphite deposit, Epanko, in Tanzania. By studying the metamorphic gradient and formation of the deposit, we determined that the rocks experienced temperatures of around 800 degrees Celsius at around 25 to 30 km deep in the crust, which is more than hot enough to make good crystal flake graphite.
This knowledge has confirmed the quality of the deposit and helped the company identify downstream processing options to produce battery grade graphite. This work is helping these companies establish a competitive edge in the market and was supported by the Australian Government’s Innovation Connections program.
You can read the full story in the in the latest edition of resourceful, which takes a closer look at the value resource characterisation is creating for the minerals industry.


Pingback: What to know about graphite: the mineral of extremes - Oresome Resources