We are testing two vaccine candidates at our Australian Centre for Disease Preparedness (formerly Australian Animal Health Laboratory or AAHL) in Geelong, Victoria.
Updated 24 April 2020
After months of research, we have a major update on our COVID-19 work. We have started pre-clinical trials for two vaccine candidates. This is happening at our Australian Centre for Disease Preparedness (ACDP) in Geelong, Victoria.
The work — in which we play a critical role — involves deep collaboration within Australia and overseas and forms part of a global rapid response to this pandemic.
Pre-clinical trials to get the vaccine faster
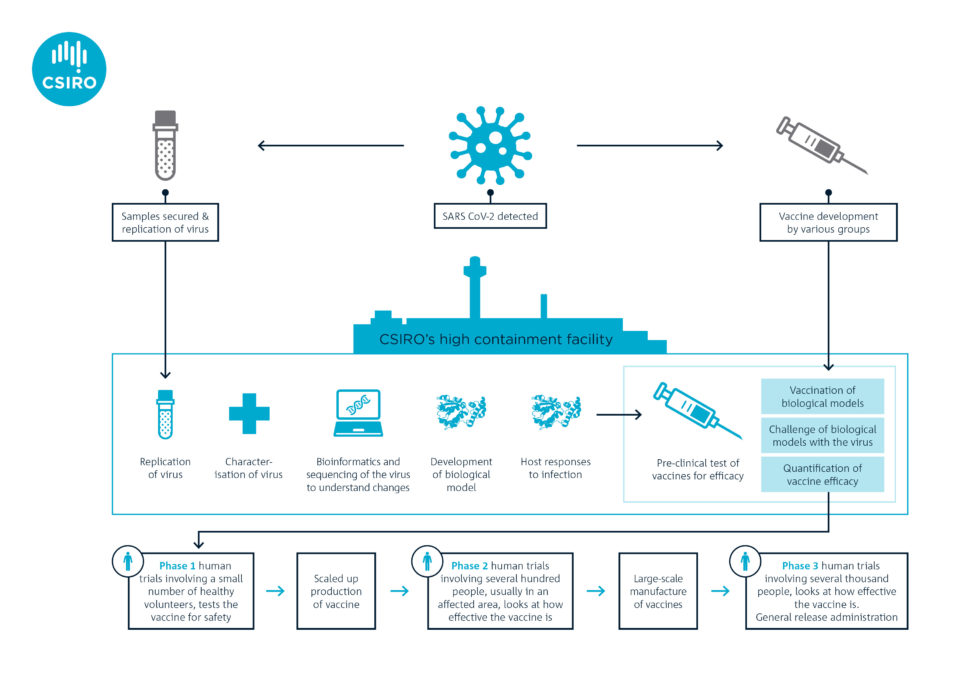
Vaccines generally take 10-15 years to develop and test. But researchers like us are hoping to streamline this process. We’re working as part of a global alliance with the Coalition for Epidemic Preparedness Innovations (CEPI). The goal is to speed up traditional vaccine development and testing and have at least one vaccine ready within 10-15 months.
CEPI asked us in January to start getting a better understanding of COVID-19 while the outbreak was in its early stages. We were the first research organisation outside of China to generate enough stocks of the virus (using the VIC01 strain isolated by the Doherty Institute) for preclinical research.
In February, we became the first team in the world to show that ferrets are susceptible to this novel coronavirus. We established the ferret model in record time and were tasked by CEPI in March to start the world’s first multi-vaccine efficacy study in animals.
In consultation with the World Health Organization (WHO), CEPI selected two candidates based on readiness for testing. The vaccine candidates come from Inovio Pharmaceuticals in the US and the University of Oxford. They’re the second and third vaccines in the world to undergo pre-clinical trials. The former is a DNA vaccine, a novel approach because there are currently no licenced DNA vaccines for humans. The latter is what’s called a vectored vaccine (an established technology).
Pre-clinical trials are underway at our high containment facility, the Australian Centre for Disease Preparedness (ACDP)
Phase 1 clinical trials
Both candidates are now entering Phase 1 clinical trials in healthy humans to see if they are safe. However, before seeking approval for Phase 2 and Phase 3 clinical trials, vaccine developers need efficacy data from pre-clinical studies. This is where organisations such as ours play a major role.
The two vaccine candidates are undergoing pre-clinical trials in our laboratory. Ferrets will be vaccinated and monitored for four weeks so it develops immunity. The ferrets will then be given a dose of the virus to measure the success of the two vaccine candidates. Such studies are planned very carefully with significant oversight of animal ethics and welfare considerations.
We’re not directly involved in developing vaccine candidates for COVID-19. But our preclinical trials are critical in helping to refine possible vaccination strategies. For example, we will test less invasive routes of vaccination (e.g. via the nose) and a two-shot regimen. The findings from our studies can have a huge impact on clinical delivery.
What else is happening at CSIRO?
We studied SARS-CoV-2’s genome sequence and confirmed the virus is changing into distinct clusters. We’re now looking at how this may impact the development and evaluation of COVID-19 vaccines, therapeutics and diagnostic tests.
In partnership with The University of Queensland, our researchers have successfully demonstrated the presence of SARS-Cov-2 in Australian untreated wastewater. This is the first step in developing an early warning surveillance system to track COVID-19 prevalence in the community.
We’re also working with the Australian Government and Victorian manufacturers to build local capability and supply of materials. This will rapidly address the demand for medical materials. And with one vaccine (developed by the University of Queensland), we are helping to scale-up production.


4th May 2020 at 3:04 pm
As always, amazing work !!!
4th May 2020 at 11:27 am
Tanks good job
2nd May 2020 at 11:35 am
Hi Kashmi,
Thank you for the above information, sounds quite promising. Can you please clarify the following;
What is the duration of phase 2 & 3 human trials, and will a variable split of gender, co morbidity, fitness, age be accounted for in your trial design? Ideally the side effects should be monitored and measured not just the efficacy, have I missed this? I don’t seem to be able to see a step in the process on monitoring for side effects? I assume this would take more than weeks to monitor, re 4 weeks animal trial, no info on duration human trials phase 2&3. – More info required
Will human trials contain the comorbidity factors prevalent in majority of deaths? Will participants be split 50/50% of ‘healthy’ and comorbidity? and than randomised? is it a double blind placebo vaccine trial? Will you account for compound variables if its not blind trial? What’s the impact on gut microbiome? neurological? cardiovascular? CNS? is this even on the radar to scope within mere weeks? (re methloquine reports psychiatric side effects on military personnel) – More info required.
Just trying to understand how this process will be undertaken, so as to feel confident in what is being produced ? Thank you
25th May 2020 at 4:09 pm
Hi there, thanks for your question.
Each vaccine developer is doing things slightly differently so unfortunately, we can’t provide the information to answer your question. Plus some of these trials are still being sorted out (due to the virus being in the areas where scientists want to test the vaccine).
However, Oxford did announce what they’re doing here https://www.ovg.ox.ac.uk/news/oxford-covid-19-vaccine-to-begin-phase-ii-iii-human-trials
This is another approach that’s being done by an Aussie developer: https://nucleusnetwork.com.au/news/novavax-to-commence-covid-19-vaccine-trial-with-nucleus-network/
Hope this helps.
Kind regards,
Kashmi
Team CSIRO
25th April 2020 at 8:22 am
My understanding is that Biotron is using their library of compounds at CSIRO for testing, as announced by the company. But no mention since has been made how their testing of their compounds has progressed and if they are assisting other organisations with this work. I thought they may have been a leader with CSIRO with available compounds ready for testing, but all has been whisper quiet. It should all be an open book as we urgently need a vaccine for covid19.
18th April 2020 at 8:19 am
Keep going – the old CSIRO at it’s best.