We are testing two vaccine candidates at our Australian Centre for Disease Preparedness (formerly Australian Animal Health Laboratory or AAHL) in Geelong, Victoria.
Updated 24 April 2020
After months of research, we have a major update on our COVID-19 work. We have started pre-clinical trials for two vaccine candidates. This is happening at our Australian Centre for Disease Preparedness (ACDP) in Geelong, Victoria.
The work — in which we play a critical role — involves deep collaboration within Australia and overseas and forms part of a global rapid response to this pandemic.
Pre-clinical trials to get the vaccine faster
Vaccines generally take 10-15 years to develop and test. But researchers like us are hoping to streamline this process. We’re working as part of a global alliance with the Coalition for Epidemic Preparedness Innovations (CEPI). The goal is to speed up traditional vaccine development and testing and have at least one vaccine ready within 10-15 months.
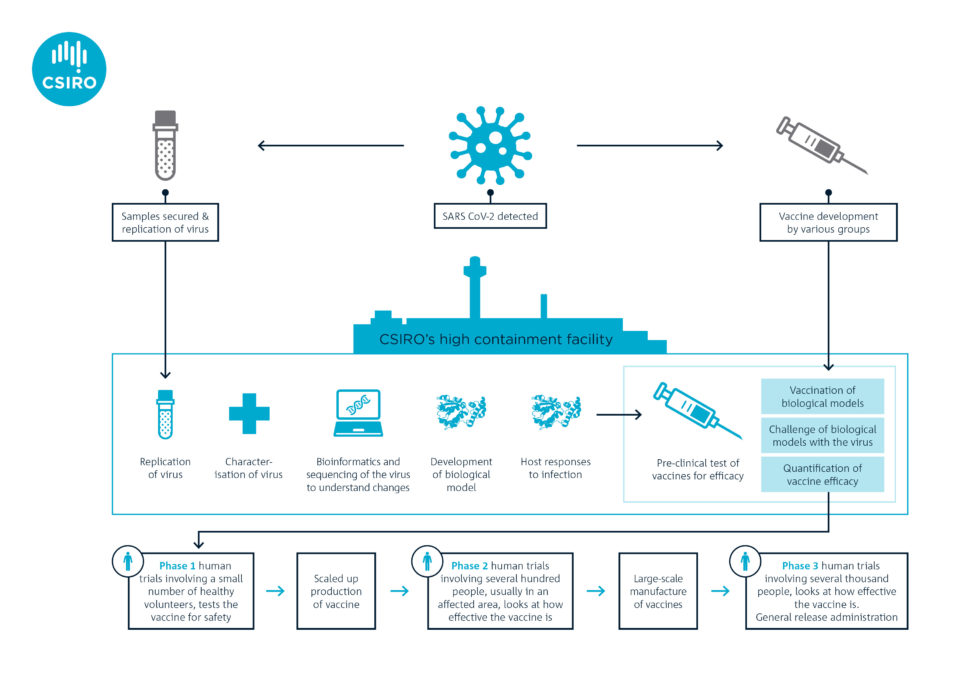
CEPI asked us in January to start getting a better understanding of COVID-19 while the outbreak was in its early stages. We were the first research organisation outside of China to generate enough stocks of the virus (using the VIC01 strain isolated by the Doherty Institute) for preclinical research.
In February, we became the first team in the world to show that ferrets are susceptible to this novel coronavirus. We established the ferret model in record time and were tasked by CEPI in March to start the world’s first multi-vaccine efficacy study in animals.
In consultation with the World Health Organization (WHO), CEPI selected two candidates based on readiness for testing. The vaccine candidates come from Inovio Pharmaceuticals in the US and the University of Oxford. They’re the second and third vaccines in the world to undergo pre-clinical trials. The former is a DNA vaccine, a novel approach because there are currently no licenced DNA vaccines for humans. The latter is what’s called a vectored vaccine (an established technology).
Pre-clinical trials are underway at our high containment facility, the Australian Centre for Disease Preparedness (ACDP)
Phase 1 clinical trials
Both candidates are now entering Phase 1 clinical trials in healthy humans to see if they are safe. However, before seeking approval for Phase 2 and Phase 3 clinical trials, vaccine developers need efficacy data from pre-clinical studies. This is where organisations such as ours play a major role.
The two vaccine candidates are undergoing pre-clinical trials in our laboratory. Ferrets will be vaccinated and monitored for four weeks so it develops immunity. The ferrets will then be given a dose of the virus to measure the success of the two vaccine candidates. Such studies are planned very carefully with significant oversight of animal ethics and welfare considerations.
We’re not directly involved in developing vaccine candidates for COVID-19. But our preclinical trials are critical in helping to refine possible vaccination strategies. For example, we will test less invasive routes of vaccination (e.g. via the nose) and a two-shot regimen. The findings from our studies can have a huge impact on clinical delivery.
What else is happening at CSIRO?
We studied SARS-CoV-2’s genome sequence and confirmed the virus is changing into distinct clusters. We’re now looking at how this may impact the development and evaluation of COVID-19 vaccines, therapeutics and diagnostic tests.
In partnership with The University of Queensland, our researchers have successfully demonstrated the presence of SARS-Cov-2 in Australian untreated wastewater. This is the first step in developing an early warning surveillance system to track COVID-19 prevalence in the community.
We’re also working with the Australian Government and Victorian manufacturers to build local capability and supply of materials. This will rapidly address the demand for medical materials. And with one vaccine (developed by the University of Queensland), we are helping to scale-up production.


16th April 2020 at 1:35 pm
Fantastic work everyone and a very honorable mention for the WHO, that does an incredible amount of work keeping us safe. Well done everyone, I wish you all the best
16th April 2020 at 12:31 pm
Keep up the good work, looking forward to seeing the results of your very important work!
16th April 2020 at 12:25 pm
Prior to covid -19 taking hold a partial preventative measure is breathing in through gauze a drop of tea tree, eucalyptus or lavendor oil through the nose, holding breath and expelling through the mouth. Sterilization and attachments of air borne particles is possible. This is no guarantee,but by using the same method COVID-19 enters the body, it is either attacked on entry or from behind directly.
Bodies heat causes complete evapourization, reaching all internal surfaces and holding breath builds up pressure ,forcing into lung tissue. Breathing out through mouth covers full circuit areas initially infected.
For your concideration
16th April 2020 at 12:11 pm
Astonishing progress in such a short time frame. The toolkit available to geneticists today was the stuff of dreams only a few years ago so although 12-18 months might seem to be a long wait, we are fortunate that Covid-19 didn’t happen even a few years earlier.
Also pleased to see some authoritative information about the state of localised mutation because there’s a lot of wild claims out there: “…the virus is evolving into a number of distinct clusters in different parts of the world. At this time, we do not think it will affect the development and evaluation of COVID-19 vaccines, therapies and diagnostics…” Fingers crossed this doesn’t change.
16th April 2020 at 12:03 pm
You may get this twice because of my computer illiteracy. The development of a usable vaccine is still a fair way off even though you hope to reduce this time to only 10% of the normal time. Would the extraction of antibodies from the blood serum of recovered COVID19 patients and injecting healthy persons be a faster, interim approach?